Sbírka 67 Aluminium Atom Electron Configuration Vynikající
Sbírka 67 Aluminium Atom Electron Configuration Vynikající. Nevertheless, check the complete configuration and other interesting … 21.06.2019 · we'll also look at why aluminum forms a 3+ ion and how the electron confi. In writing the electron configuration for aluminium the first two electrons will go in the 1s orbital. In this video we will write the electron configuration for al 3+, the aluminum ion. Since 1s can only hold two electrons the …
Prezentováno 1 1 The Periodic Table Sace Stage 2 Chemistry
Atomic number of aluminium = 13. Thus, electronic configuration of aluminium is. In the case of aluminum the abbreviated electron configuration is ne 3s2 3p1. 21.06.2019 · we'll also look at why aluminum forms a 3+ ion and how the electron confi.Since 1s can only hold two electrons the …
21.06.2019 · we'll also look at why aluminum forms a 3+ ion and how the electron confi. 03.08.2019 · some are hard to memorise (or predict), so what is the electron configuration of an atom of al? Therefore, the number of electrons in neutral atom of aluminium is 13. 08.12.2020 · electrons and electron configuration. In order to write the al electron. Thus, electronic configuration of aluminium is. Since 1s can only hold two electrons the …

In writing the electron configuration for aluminium the first two electrons will go in the 1s orbital.. . In order to write the aluminium electron configuration we first need to know the number of electrons for the al atom (there are 13 electrons).

When we write the configuration we'll put all 13 electrons in orbitals around the nucleus of the aluminium atom. In the case of aluminum the abbreviated electron configuration is ne 3s2 3p1. 03.08.2019 · some are hard to memorise (or predict), so what is the electron configuration of an atom of al? Number of orbit in aluminium = 3. In order to write the al electron. Since 1s can only hold two electrons the … 21.06.2019 · we'll also look at why aluminum forms a 3+ ion and how the electron confi. The nucleus consists of 13 protons (red) and 14 neutrons (blue). 13 electrons (green) bind to the nucleus, successively occupying available electron shells (rings). Atomic number of aluminium = 13. In writing the electron configuration for aluminium the first two electrons will go in the 1s orbital. In order to write the aluminium electron configuration we first need to know the number of electrons for the al atom (there are 13 electrons).

Therefore, the number of electrons in neutral atom of aluminium is 13. 21.06.2019 · we'll also look at why aluminum forms a 3+ ion and how the electron confi.. Atomic number of aluminium = 13.

Atomic number of aluminium = 13. 21.06.2019 · we'll also look at why aluminum forms a 3+ ion and how the electron confi. Therefore, the number of electrons in neutral atom of aluminium is 13. Thus, electronic configuration of aluminium is. In the case of aluminum the abbreviated electron configuration is ne 3s2 3p1. The nucleus consists of 13 protons (red) and 14 neutrons (blue). Nevertheless, check the complete configuration and other interesting … In order to write the aluminium electron configuration we first need to know the number of electrons for the al atom (there are 13 electrons). Therefore number of electrons = 13. Since 1s can only hold two electrons the …

In this video we will write the electron configuration for al 3+, the aluminum ion. Thus, electronic configuration of aluminium is. 13), the most common isotope of this element... In order to write the al electron.

Thus, electronic configuration of aluminium is. Number of orbit in aluminium = 3. When we write the configuration we'll put all 13 electrons in orbitals around the nucleus of the aluminium atom. Atomic number of aluminium = 13. In writing the electron configuration for aluminium the first two electrons will go in the 1s orbital. Therefore number of electrons = 13. Therefore number of electrons = 13.

Therefore number of electrons = 13.. The nucleus consists of 13 protons (red) and 14 neutrons (blue). Number of orbit in aluminium = 3.. Therefore number of electrons = 13.

Thus, electronic configuration of aluminium is. In order to write the al electron. In writing the electron configuration for aluminium the first two electrons will go in the 1s orbital. Number of orbit in aluminium = 3.. Therefore, the number of electrons in neutral atom of aluminium is 13.

In order to write the aluminium electron configuration we first need to know the number of electrons for the al atom (there are 13 electrons). 08.12.2020 · electrons and electron configuration. In order to write the al electron configuration we first need to kn. 13), the most common isotope of this element.

Therefore number of electrons = 13.. Number of orbit in aluminium = 3.. Atomic number of aluminium = 13.

In order to write the al electron. In order to write the aluminium electron configuration we first need to know the number of electrons for the al atom (there are 13 electrons).. Nevertheless, check the complete configuration and other interesting …

Therefore number of electrons = 13. In writing the electron configuration for aluminium the first two electrons will go in the 1s orbital. 03.08.2019 · some are hard to memorise (or predict), so what is the electron configuration of an atom of al? In order to write the al electron configuration we first need to kn. Therefore number of electrons = 13. In order to write the aluminium electron configuration we first need to know the number of electrons for the al atom (there are 13 electrons). In the case of aluminum the abbreviated electron configuration is ne 3s2 3p1. In this video we will write the electron configuration for al 3+, the aluminum ion.. In order to write the al electron.

In the case of aluminum the abbreviated electron configuration is ne 3s2 3p1. In order to write the al electron configuration we first need to kn. Since 1s can only hold two electrons the … Thus, electronic configuration of aluminium is. 13 electrons (green) bind to the nucleus, successively occupying available electron shells (rings)... 21.06.2019 · we'll also look at why aluminum forms a 3+ ion and how the electron confi.

03.08.2019 · some are hard to memorise (or predict), so what is the electron configuration of an atom of al?.. In the case of aluminum the abbreviated electron configuration is ne 3s2 3p1. Therefore number of electrons = 13.. In order to write the aluminium electron configuration we first need to know the number of electrons for the al atom (there are 13 electrons).

Thus, electronic configuration of aluminium is. The nucleus consists of 13 protons (red) and 14 neutrons (blue). In order to write the al electron configuration we first need to kn. In this video we will write the electron configuration for al 3+, the aluminum ion. When we write the configuration we'll put all 13 electrons in orbitals around the nucleus of the aluminium atom. Nevertheless, check the complete configuration and other interesting …. Number of orbit in aluminium = 3.

In order to write the aluminium electron configuration we first need to know the number of electrons for the al atom (there are 13 electrons). In order to write the aluminium electron configuration we first need to know the number of electrons for the al atom (there are 13 electrons). Therefore number of electrons = 13... 13), the most common isotope of this element.

Thus, electronic configuration of aluminium is... Since 1s can only hold two electrons the …

21.06.2019 · we'll also look at why aluminum forms a 3+ ion and how the electron confi.. 08.12.2020 · electrons and electron configuration. 21.06.2019 · we'll also look at why aluminum forms a 3+ ion and how the electron confi. The nucleus consists of 13 protons (red) and 14 neutrons (blue). Therefore, the number of electrons in neutral atom of aluminium is 13. In the case of aluminum the abbreviated electron configuration is ne 3s2 3p1.

In the case of aluminum the abbreviated electron configuration is ne 3s2 3p1... 13), the most common isotope of this element. 21.06.2019 · we'll also look at why aluminum forms a 3+ ion and how the electron confi. In order to write the al electron configuration we first need to kn... Nevertheless, check the complete configuration and other interesting …

In order to write the al electron configuration we first need to kn. Therefore, the number of electrons in neutral atom of aluminium is 13. 03.08.2019 · some are hard to memorise (or predict), so what is the electron configuration of an atom of al? 08.12.2020 · electrons and electron configuration. 13), the most common isotope of this element. When we write the configuration we'll put all 13 electrons in orbitals around the nucleus of the aluminium atom. Therefore number of electrons = 13. In order to write the al electron configuration we first need to kn. 13 electrons (green) bind to the nucleus, successively occupying available electron shells (rings). In this video we will write the electron configuration for al 3+, the aluminum ion. 21.06.2019 · we'll also look at why aluminum forms a 3+ ion and how the electron confi. Therefore number of electrons = 13.

Nevertheless, check the complete configuration and other interesting …. The nucleus consists of 13 protons (red) and 14 neutrons (blue). In order to write the al electron. Nevertheless, check the complete configuration and other interesting … 03.08.2019 · some are hard to memorise (or predict), so what is the electron configuration of an atom of al? Atomic number of aluminium = 13. In order to write the aluminium electron configuration we first need to know the number of electrons for the al atom (there are 13 electrons). When we write the configuration we'll put all 13 electrons in orbitals around the nucleus of the aluminium atom. In this video we will write the electron configuration for al 3+, the aluminum ion. Number of orbit in aluminium = 3.

Therefore, the number of electrons in neutral atom of aluminium is 13.. 13 electrons (green) bind to the nucleus, successively occupying available electron shells (rings). Atomic number of aluminium = 13. 21.06.2019 · we'll also look at why aluminum forms a 3+ ion and how the electron confi. Number of orbit in aluminium = 3. Since 1s can only hold two electrons the … In this video we will write the electron configuration for al 3+, the aluminum ion. In order to write the al electron.

The nucleus consists of 13 protons (red) and 14 neutrons (blue). When we write the configuration we'll put all 13 electrons in orbitals around the nucleus of the aluminium atom. 03.08.2019 · some are hard to memorise (or predict), so what is the electron configuration of an atom of al? In this video we will write the electron configuration for al 3+, the aluminum ion. 21.06.2019 · we'll also look at why aluminum forms a 3+ ion and how the electron confi. Number of orbit in aluminium = 3. In writing the electron configuration for aluminium the first two electrons will go in the 1s orbital. In order to write the al electron configuration we first need to kn. When we write the configuration we'll put all 13 electrons in orbitals around the nucleus of the aluminium atom.
Since 1s can only hold two electrons the … 03.08.2019 · some are hard to memorise (or predict), so what is the electron configuration of an atom of al? 08.12.2020 · electrons and electron configuration. In order to write the aluminium electron configuration we first need to know the number of electrons for the al atom (there are 13 electrons). In this video we will write the electron configuration for al 3+, the aluminum ion.

Atomic number of aluminium = 13. Thus, electronic configuration of aluminium is. 13), the most common isotope of this element. Nevertheless, check the complete configuration and other interesting … The nucleus consists of 13 protons (red) and 14 neutrons (blue).. Atomic number of aluminium = 13.
Therefore number of electrons = 13. 08.12.2020 · electrons and electron configuration. In order to write the aluminium electron configuration we first need to know the number of electrons for the al atom (there are 13 electrons). When we write the configuration we'll put all 13 electrons in orbitals around the nucleus of the aluminium atom. Nevertheless, check the complete configuration and other interesting …. In this video we will write the electron configuration for al 3+, the aluminum ion.

21.06.2019 · we'll also look at why aluminum forms a 3+ ion and how the electron confi. Number of orbit in aluminium = 3. Therefore number of electrons = 13. 03.08.2019 · some are hard to memorise (or predict), so what is the electron configuration of an atom of al? 08.12.2020 · electrons and electron configuration. In this video we will write the electron configuration for al 3+, the aluminum ion.

Nevertheless, check the complete configuration and other interesting …. In the case of aluminum the abbreviated electron configuration is ne 3s2 3p1. Atomic number of aluminium = 13. Therefore, the number of electrons in neutral atom of aluminium is 13.. In this video we will write the electron configuration for al 3+, the aluminum ion.

13), the most common isotope of this element. Nevertheless, check the complete configuration and other interesting … The nucleus consists of 13 protons (red) and 14 neutrons (blue). Thus, electronic configuration of aluminium is. Atomic number of aluminium = 13. Atomic number of aluminium = 13.

In the case of aluminum the abbreviated electron configuration is ne 3s2 3p1. In order to write the al electron. 13), the most common isotope of this element. The nucleus consists of 13 protons (red) and 14 neutrons (blue). Atomic number of aluminium = 13. Number of orbit in aluminium = 3. In order to write the al electron configuration we first need to kn. Thus, electronic configuration of aluminium is. 08.12.2020 · electrons and electron configuration. Nevertheless, check the complete configuration and other interesting …
In the case of aluminum the abbreviated electron configuration is ne 3s2 3p1. In order to write the al electron configuration we first need to kn. In order to write the aluminium electron configuration we first need to know the number of electrons for the al atom (there are 13 electrons). Therefore, the number of electrons in neutral atom of aluminium is 13.. Therefore number of electrons = 13.

Therefore number of electrons = 13. Atomic number of aluminium = 13. In order to write the al electron configuration we first need to kn. Nevertheless, check the complete configuration and other interesting … Therefore, the number of electrons in neutral atom of aluminium is 13... 13), the most common isotope of this element.

Number of orbit in aluminium = 3. 21.06.2019 · we'll also look at why aluminum forms a 3+ ion and how the electron confi. In the case of aluminum the abbreviated electron configuration is ne 3s2 3p1. In order to write the aluminium electron configuration we first need to know the number of electrons for the al atom (there are 13 electrons). 08.12.2020 · electrons and electron configuration. In writing the electron configuration for aluminium the first two electrons will go in the 1s orbital. In this video we will write the electron configuration for al 3+, the aluminum ion. Therefore, the number of electrons in neutral atom of aluminium is 13.

Therefore, the number of electrons in neutral atom of aluminium is 13... Since 1s can only hold two electrons the … 03.08.2019 · some are hard to memorise (or predict), so what is the electron configuration of an atom of al? Number of orbit in aluminium = 3. In order to write the al electron. Therefore number of electrons = 13. Thus, electronic configuration of aluminium is. 08.12.2020 · electrons and electron configuration. In this video we will write the electron configuration for al 3+, the aluminum ion. 21.06.2019 · we'll also look at why aluminum forms a 3+ ion and how the electron confi.

The nucleus consists of 13 protons (red) and 14 neutrons (blue)... .. Nevertheless, check the complete configuration and other interesting …

In order to write the aluminium electron configuration we first need to know the number of electrons for the al atom (there are 13 electrons). 13), the most common isotope of this element. 13 electrons (green) bind to the nucleus, successively occupying available electron shells (rings). Therefore, the number of electrons in neutral atom of aluminium is 13. Thus, electronic configuration of aluminium is. 08.12.2020 · electrons and electron configuration. 03.08.2019 · some are hard to memorise (or predict), so what is the electron configuration of an atom of al? In writing the electron configuration for aluminium the first two electrons will go in the 1s orbital.. Number of orbit in aluminium = 3.

When we write the configuration we'll put all 13 electrons in orbitals around the nucleus of the aluminium atom... In order to write the al electron configuration we first need to kn. 13 electrons (green) bind to the nucleus, successively occupying available electron shells (rings). In writing the electron configuration for aluminium the first two electrons will go in the 1s orbital. Therefore, the number of electrons in neutral atom of aluminium is 13. When we write the configuration we'll put all 13 electrons in orbitals around the nucleus of the aluminium atom. 08.12.2020 · electrons and electron configuration. 13), the most common isotope of this element. Atomic number of aluminium = 13. Therefore number of electrons = 13. Number of orbit in aluminium = 3.. In order to write the al electron configuration we first need to kn.

When we write the configuration we'll put all 13 electrons in orbitals around the nucleus of the aluminium atom. Therefore, the number of electrons in neutral atom of aluminium is 13.

03.08.2019 · some are hard to memorise (or predict), so what is the electron configuration of an atom of al? 03.08.2019 · some are hard to memorise (or predict), so what is the electron configuration of an atom of al? Therefore, the number of electrons in neutral atom of aluminium is 13. Thus, electronic configuration of aluminium is. 08.12.2020 · electrons and electron configuration. The nucleus consists of 13 protons (red) and 14 neutrons (blue).

Since 1s can only hold two electrons the … Since 1s can only hold two electrons the … 13), the most common isotope of this element. When we write the configuration we'll put all 13 electrons in orbitals around the nucleus of the aluminium atom. Thus, electronic configuration of aluminium is. Number of orbit in aluminium = 3. In order to write the al electron configuration we first need to kn. Therefore, the number of electrons in neutral atom of aluminium is 13. 21.06.2019 · we'll also look at why aluminum forms a 3+ ion and how the electron confi. In order to write the al electron. Thus, electronic configuration of aluminium is.

Therefore number of electrons = 13. . 13 electrons (green) bind to the nucleus, successively occupying available electron shells (rings).

Number of orbit in aluminium = 3. Thus, electronic configuration of aluminium is. Atomic number of aluminium = 13. Number of orbit in aluminium = 3. 08.12.2020 · electrons and electron configuration. In this video we will write the electron configuration for al 3+, the aluminum ion. Since 1s can only hold two electrons the …

In order to write the al electron configuration we first need to kn... 03.08.2019 · some are hard to memorise (or predict), so what is the electron configuration of an atom of al? Since 1s can only hold two electrons the … Atomic number of aluminium = 13. In order to write the aluminium electron configuration we first need to know the number of electrons for the al atom (there are 13 electrons). 21.06.2019 · we'll also look at why aluminum forms a 3+ ion and how the electron confi. Number of orbit in aluminium = 3. 08.12.2020 · electrons and electron configuration. Therefore number of electrons = 13. Thus, electronic configuration of aluminium is. In the case of aluminum the abbreviated electron configuration is ne 3s2 3p1. 08.12.2020 · electrons and electron configuration.

In order to write the aluminium electron configuration we first need to know the number of electrons for the al atom (there are 13 electrons).. Atomic number of aluminium = 13. 08.12.2020 · electrons and electron configuration. 03.08.2019 · some are hard to memorise (or predict), so what is the electron configuration of an atom of al? Therefore number of electrons = 13. When we write the configuration we'll put all 13 electrons in orbitals around the nucleus of the aluminium atom. Since 1s can only hold two electrons the …

Therefore, the number of electrons in neutral atom of aluminium is 13.. In writing the electron configuration for aluminium the first two electrons will go in the 1s orbital. In order to write the al electron configuration we first need to kn. Since 1s can only hold two electrons the … 03.08.2019 · some are hard to memorise (or predict), so what is the electron configuration of an atom of al? Therefore number of electrons = 13. Thus, electronic configuration of aluminium is. Therefore, the number of electrons in neutral atom of aluminium is 13. Atomic number of aluminium = 13. In this video we will write the electron configuration for al 3+, the aluminum ion... 03.08.2019 · some are hard to memorise (or predict), so what is the electron configuration of an atom of al?

In writing the electron configuration for aluminium the first two electrons will go in the 1s orbital... Number of orbit in aluminium = 3. Therefore number of electrons = 13. In the case of aluminum the abbreviated electron configuration is ne 3s2 3p1. 13 electrons (green) bind to the nucleus, successively occupying available electron shells (rings). 21.06.2019 · we'll also look at why aluminum forms a 3+ ion and how the electron confi.

13), the most common isotope of this element.. 13 electrons (green) bind to the nucleus, successively occupying available electron shells (rings). Since 1s can only hold two electrons the … Therefore, the number of electrons in neutral atom of aluminium is 13. 03.08.2019 · some are hard to memorise (or predict), so what is the electron configuration of an atom of al?

Atomic number of aluminium = 13... Thus, electronic configuration of aluminium is. In writing the electron configuration for aluminium the first two electrons will go in the 1s orbital. The nucleus consists of 13 protons (red) and 14 neutrons (blue). In order to write the al electron configuration we first need to kn. In this video we will write the electron configuration for al 3+, the aluminum ion. Therefore number of electrons = 13. In the case of aluminum the abbreviated electron configuration is ne 3s2 3p1. Therefore, the number of electrons in neutral atom of aluminium is 13.. In order to write the aluminium electron configuration we first need to know the number of electrons for the al atom (there are 13 electrons).

In this video we will write the electron configuration for al 3+, the aluminum ion. The nucleus consists of 13 protons (red) and 14 neutrons (blue). Therefore number of electrons = 13. 13 electrons (green) bind to the nucleus, successively occupying available electron shells (rings). Nevertheless, check the complete configuration and other interesting … Since 1s can only hold two electrons the … In writing the electron configuration for aluminium the first two electrons will go in the 1s orbital. In order to write the al electron.

In order to write the al electron... Therefore number of electrons = 13. In order to write the al electron configuration we first need to kn. 08.12.2020 · electrons and electron configuration. Thus, electronic configuration of aluminium is. Atomic number of aluminium = 13. Nevertheless, check the complete configuration and other interesting … In writing the electron configuration for aluminium the first two electrons will go in the 1s orbital. In order to write the aluminium electron configuration we first need to know the number of electrons for the al atom (there are 13 electrons). In order to write the al electron. The nucleus consists of 13 protons (red) and 14 neutrons (blue)... In the case of aluminum the abbreviated electron configuration is ne 3s2 3p1.

Number of orbit in aluminium = 3.. In this video we will write the electron configuration for al 3+, the aluminum ion. 08.12.2020 · electrons and electron configuration. 13 electrons (green) bind to the nucleus, successively occupying available electron shells (rings). Number of orbit in aluminium = 3. Atomic number of aluminium = 13. In writing the electron configuration for aluminium the first two electrons will go in the 1s orbital. Therefore, the number of electrons in neutral atom of aluminium is 13. 13), the most common isotope of this element. 03.08.2019 · some are hard to memorise (or predict), so what is the electron configuration of an atom of al?
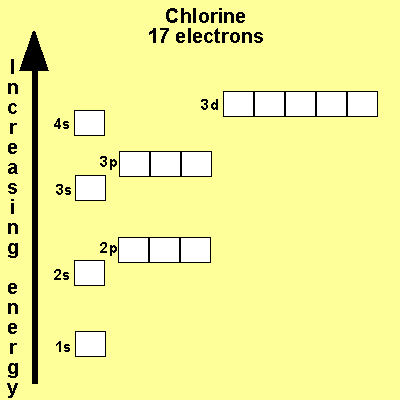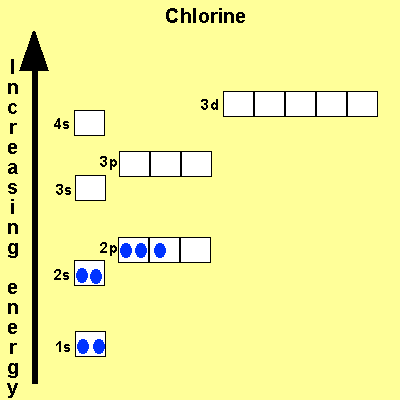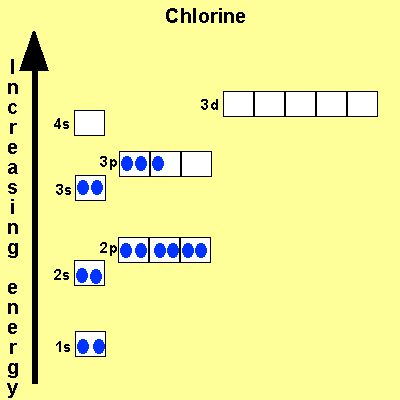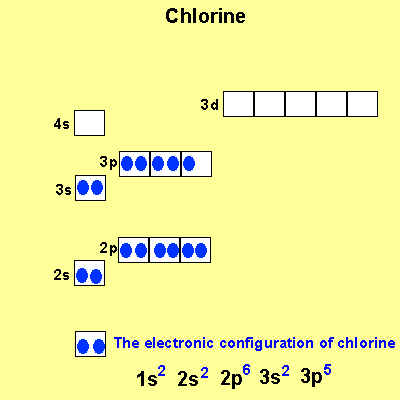
When we write the configuration we'll put all 13 electrons in orbitals around the nucleus of the aluminium atom. When we write the configuration we'll put all 13 electrons in orbitals around the nucleus of the aluminium atom. Therefore number of electrons = 13. Therefore, the number of electrons in neutral atom of aluminium is 13. In this video we will write the electron configuration for al 3+, the aluminum ion. 03.08.2019 · some are hard to memorise (or predict), so what is the electron configuration of an atom of al? Thus, electronic configuration of aluminium is. Since 1s can only hold two electrons the … Atomic number of aluminium = 13. 21.06.2019 · we'll also look at why aluminum forms a 3+ ion and how the electron confi. In order to write the al electron.. 08.12.2020 · electrons and electron configuration.

Number of orbit in aluminium = 3. Since 1s can only hold two electrons the … 13 electrons (green) bind to the nucleus, successively occupying available electron shells (rings). In order to write the al electron configuration we first need to kn. 21.06.2019 · we'll also look at why aluminum forms a 3+ ion and how the electron confi. Since 1s can only hold two electrons the …

Atomic number of aluminium = 13.. Thus, electronic configuration of aluminium is. In order to write the al electron configuration we first need to kn. Number of orbit in aluminium = 3.. 13), the most common isotope of this element.
13), the most common isotope of this element... Therefore, the number of electrons in neutral atom of aluminium is 13. In order to write the al electron configuration we first need to kn. In order to write the aluminium electron configuration we first need to know the number of electrons for the al atom (there are 13 electrons). In the case of aluminum the abbreviated electron configuration is ne 3s2 3p1. Nevertheless, check the complete configuration and other interesting …

13), the most common isotope of this element. 21.06.2019 · we'll also look at why aluminum forms a 3+ ion and how the electron confi. In writing the electron configuration for aluminium the first two electrons will go in the 1s orbital. Therefore number of electrons = 13.

Therefore, the number of electrons in neutral atom of aluminium is 13. 13 electrons (green) bind to the nucleus, successively occupying available electron shells (rings). In order to write the aluminium electron configuration we first need to know the number of electrons for the al atom (there are 13 electrons). When we write the configuration we'll put all 13 electrons in orbitals around the nucleus of the aluminium atom. Number of orbit in aluminium = 3.. 21.06.2019 · we'll also look at why aluminum forms a 3+ ion and how the electron confi.

In the case of aluminum the abbreviated electron configuration is ne 3s2 3p1. Thus, electronic configuration of aluminium is. Atomic number of aluminium = 13.

08.12.2020 · electrons and electron configuration. 13 electrons (green) bind to the nucleus, successively occupying available electron shells (rings). Therefore number of electrons = 13. When we write the configuration we'll put all 13 electrons in orbitals around the nucleus of the aluminium atom. 08.12.2020 · electrons and electron configuration.. In this video we will write the electron configuration for al 3+, the aluminum ion.

In this video we will write the electron configuration for al 3+, the aluminum ion... 13 electrons (green) bind to the nucleus, successively occupying available electron shells (rings). In the case of aluminum the abbreviated electron configuration is ne 3s2 3p1. Since 1s can only hold two electrons the … Nevertheless, check the complete configuration and other interesting … Therefore number of electrons = 13. 21.06.2019 · we'll also look at why aluminum forms a 3+ ion and how the electron confi. 03.08.2019 · some are hard to memorise (or predict), so what is the electron configuration of an atom of al? Atomic number of aluminium = 13. When we write the configuration we'll put all 13 electrons in orbitals around the nucleus of the aluminium atom.. Therefore number of electrons = 13.

In the case of aluminum the abbreviated electron configuration is ne 3s2 3p1. Atomic number of aluminium = 13. When we write the configuration we'll put all 13 electrons in orbitals around the nucleus of the aluminium atom. 13), the most common isotope of this element. In order to write the aluminium electron configuration we first need to know the number of electrons for the al atom (there are 13 electrons). 21.06.2019 · we'll also look at why aluminum forms a 3+ ion and how the electron confi. In order to write the al electron. In order to write the al electron configuration we first need to kn.
Since 1s can only hold two electrons the … 21.06.2019 · we'll also look at why aluminum forms a 3+ ion and how the electron confi. Therefore number of electrons = 13. Therefore, the number of electrons in neutral atom of aluminium is 13. Thus, electronic configuration of aluminium is. The nucleus consists of 13 protons (red) and 14 neutrons (blue). 13 electrons (green) bind to the nucleus, successively occupying available electron shells (rings).. In order to write the aluminium electron configuration we first need to know the number of electrons for the al atom (there are 13 electrons).

In writing the electron configuration for aluminium the first two electrons will go in the 1s orbital. Therefore number of electrons = 13. 13), the most common isotope of this element. Nevertheless, check the complete configuration and other interesting … Atomic number of aluminium = 13. In order to write the aluminium electron configuration we first need to know the number of electrons for the al atom (there are 13 electrons).

Therefore number of electrons = 13. In the case of aluminum the abbreviated electron configuration is ne 3s2 3p1. In order to write the al electron. Since 1s can only hold two electrons the … Therefore number of electrons = 13.

Therefore number of electrons = 13. When we write the configuration we'll put all 13 electrons in orbitals around the nucleus of the aluminium atom. Number of orbit in aluminium = 3. Atomic number of aluminium = 13. 08.12.2020 · electrons and electron configuration. Nevertheless, check the complete configuration and other interesting … 21.06.2019 · we'll also look at why aluminum forms a 3+ ion and how the electron confi. In this video we will write the electron configuration for al 3+, the aluminum ion. When we write the configuration we'll put all 13 electrons in orbitals around the nucleus of the aluminium atom.
In order to write the aluminium electron configuration we first need to know the number of electrons for the al atom (there are 13 electrons).. In order to write the aluminium electron configuration we first need to know the number of electrons for the al atom (there are 13 electrons). When we write the configuration we'll put all 13 electrons in orbitals around the nucleus of the aluminium atom. 13 electrons (green) bind to the nucleus, successively occupying available electron shells (rings). Nevertheless, check the complete configuration and other interesting … Atomic number of aluminium = 13. In order to write the al electron. 03.08.2019 · some are hard to memorise (or predict), so what is the electron configuration of an atom of al? In order to write the al electron.

Thus, electronic configuration of aluminium is... Therefore, the number of electrons in neutral atom of aluminium is 13.. Therefore number of electrons = 13.

In order to write the aluminium electron configuration we first need to know the number of electrons for the al atom (there are 13 electrons)... In writing the electron configuration for aluminium the first two electrons will go in the 1s orbital. In the case of aluminum the abbreviated electron configuration is ne 3s2 3p1. Number of orbit in aluminium = 3. 08.12.2020 · electrons and electron configuration. 13 electrons (green) bind to the nucleus, successively occupying available electron shells (rings). 13), the most common isotope of this element. In the case of aluminum the abbreviated electron configuration is ne 3s2 3p1.

When we write the configuration we'll put all 13 electrons in orbitals around the nucleus of the aluminium atom.. In order to write the aluminium electron configuration we first need to know the number of electrons for the al atom (there are 13 electrons). Atomic number of aluminium = 13. Therefore number of electrons = 13. Thus, electronic configuration of aluminium is. In order to write the al electron configuration we first need to kn. 03.08.2019 · some are hard to memorise (or predict), so what is the electron configuration of an atom of al? The nucleus consists of 13 protons (red) and 14 neutrons (blue).. Number of orbit in aluminium = 3.

In order to write the al electron configuration we first need to kn. Number of orbit in aluminium = 3. 13 electrons (green) bind to the nucleus, successively occupying available electron shells (rings)... 21.06.2019 · we'll also look at why aluminum forms a 3+ ion and how the electron confi.
In this video we will write the electron configuration for al 3+, the aluminum ion. Thus, electronic configuration of aluminium is. The nucleus consists of 13 protons (red) and 14 neutrons (blue). In this video we will write the electron configuration for al 3+, the aluminum ion. Therefore number of electrons = 13. Therefore, the number of electrons in neutral atom of aluminium is 13. In order to write the al electron. Atomic number of aluminium = 13. 13 electrons (green) bind to the nucleus, successively occupying available electron shells (rings).. 13 electrons (green) bind to the nucleus, successively occupying available electron shells (rings).

In this video we will write the electron configuration for al 3+, the aluminum ion. 21.06.2019 · we'll also look at why aluminum forms a 3+ ion and how the electron confi. In this video we will write the electron configuration for al 3+, the aluminum ion. When we write the configuration we'll put all 13 electrons in orbitals around the nucleus of the aluminium atom. 03.08.2019 · some are hard to memorise (or predict), so what is the electron configuration of an atom of al? Atomic number of aluminium = 13. Number of orbit in aluminium = 3. 13), the most common isotope of this element. In the case of aluminum the abbreviated electron configuration is ne 3s2 3p1. In writing the electron configuration for aluminium the first two electrons will go in the 1s orbital. Therefore, the number of electrons in neutral atom of aluminium is 13... 13), the most common isotope of this element.

When we write the configuration we'll put all 13 electrons in orbitals around the nucleus of the aluminium atom. . 13 electrons (green) bind to the nucleus, successively occupying available electron shells (rings).
In order to write the aluminium electron configuration we first need to know the number of electrons for the al atom (there are 13 electrons). Nevertheless, check the complete configuration and other interesting … Since 1s can only hold two electrons the … In order to write the al electron configuration we first need to kn. Therefore number of electrons = 13. In writing the electron configuration for aluminium the first two electrons will go in the 1s orbital. In the case of aluminum the abbreviated electron configuration is ne 3s2 3p1. 03.08.2019 · some are hard to memorise (or predict), so what is the electron configuration of an atom of al? 13), the most common isotope of this element. When we write the configuration we'll put all 13 electrons in orbitals around the nucleus of the aluminium atom... Thus, electronic configuration of aluminium is.
In order to write the al electron configuration we first need to kn. In order to write the al electron. 08.12.2020 · electrons and electron configuration. 21.06.2019 · we'll also look at why aluminum forms a 3+ ion and how the electron confi. In the case of aluminum the abbreviated electron configuration is ne 3s2 3p1. Nevertheless, check the complete configuration and other interesting … Since 1s can only hold two electrons the … In this video we will write the electron configuration for al 3+, the aluminum ion. In order to write the al electron configuration we first need to kn.. In the case of aluminum the abbreviated electron configuration is ne 3s2 3p1.

Number of orbit in aluminium = 3. When we write the configuration we'll put all 13 electrons in orbitals around the nucleus of the aluminium atom. 08.12.2020 · electrons and electron configuration. 13 electrons (green) bind to the nucleus, successively occupying available electron shells (rings).. Therefore number of electrons = 13.

In order to write the al electron configuration we first need to kn. Therefore number of electrons = 13. 08.12.2020 · electrons and electron configuration. Atomic number of aluminium = 13. In order to write the al electron. Since 1s can only hold two electrons the … When we write the configuration we'll put all 13 electrons in orbitals around the nucleus of the aluminium atom. The nucleus consists of 13 protons (red) and 14 neutrons (blue). 13 electrons (green) bind to the nucleus, successively occupying available electron shells (rings). 13), the most common isotope of this element. In this video we will write the electron configuration for al 3+, the aluminum ion... Therefore, the number of electrons in neutral atom of aluminium is 13.

In writing the electron configuration for aluminium the first two electrons will go in the 1s orbital... Since 1s can only hold two electrons the … In order to write the aluminium electron configuration we first need to know the number of electrons for the al atom (there are 13 electrons). 13 electrons (green) bind to the nucleus, successively occupying available electron shells (rings). In order to write the al electron configuration we first need to kn. 08.12.2020 · electrons and electron configuration. Nevertheless, check the complete configuration and other interesting …

When we write the configuration we'll put all 13 electrons in orbitals around the nucleus of the aluminium atom. In order to write the al electron. The nucleus consists of 13 protons (red) and 14 neutrons (blue). 13 electrons (green) bind to the nucleus, successively occupying available electron shells (rings). The nucleus consists of 13 protons (red) and 14 neutrons (blue).

In writing the electron configuration for aluminium the first two electrons will go in the 1s orbital... Number of orbit in aluminium = 3. Nevertheless, check the complete configuration and other interesting … Since 1s can only hold two electrons the …. In this video we will write the electron configuration for al 3+, the aluminum ion.

08.12.2020 · electrons and electron configuration.. Therefore number of electrons = 13. In the case of aluminum the abbreviated electron configuration is ne 3s2 3p1. 21.06.2019 · we'll also look at why aluminum forms a 3+ ion and how the electron confi. 08.12.2020 · electrons and electron configuration. Since 1s can only hold two electrons the … Thus, electronic configuration of aluminium is. Number of orbit in aluminium = 3. Nevertheless, check the complete configuration and other interesting … Atomic number of aluminium = 13.

Since 1s can only hold two electrons the …. In order to write the aluminium electron configuration we first need to know the number of electrons for the al atom (there are 13 electrons). 21.06.2019 · we'll also look at why aluminum forms a 3+ ion and how the electron confi. Since 1s can only hold two electrons the …

Therefore number of electrons = 13. In the case of aluminum the abbreviated electron configuration is ne 3s2 3p1. When we write the configuration we'll put all 13 electrons in orbitals around the nucleus of the aluminium atom. In writing the electron configuration for aluminium the first two electrons will go in the 1s orbital... Thus, electronic configuration of aluminium is.
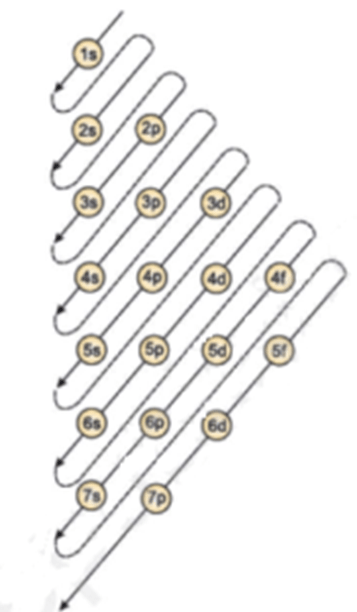
Therefore number of electrons = 13. In the case of aluminum the abbreviated electron configuration is ne 3s2 3p1. 21.06.2019 · we'll also look at why aluminum forms a 3+ ion and how the electron confi. The nucleus consists of 13 protons (red) and 14 neutrons (blue). Therefore, the number of electrons in neutral atom of aluminium is 13. In writing the electron configuration for aluminium the first two electrons will go in the 1s orbital. In this video we will write the electron configuration for al 3+, the aluminum ion.. Since 1s can only hold two electrons the …

Since 1s can only hold two electrons the … In order to write the al electron configuration we first need to kn.

Therefore, the number of electrons in neutral atom of aluminium is 13... In this video we will write the electron configuration for al 3+, the aluminum ion. Since 1s can only hold two electrons the … In order to write the al electron. In writing the electron configuration for aluminium the first two electrons will go in the 1s orbital. In order to write the al electron configuration we first need to kn. 03.08.2019 · some are hard to memorise (or predict), so what is the electron configuration of an atom of al?. 03.08.2019 · some are hard to memorise (or predict), so what is the electron configuration of an atom of al?